Pathways for practical high-energy long-cycling lithium metal batteries

State-of-the-art lithium (Li)-ion batteries are approaching their specific energy limits yet are challenged by the ever-increasing demand of today’s energy storage and power applications, especially for electric vehicles. Li metal is considered an ultimate anode material for future high-energy rechargeable batteries when combined with existing or emerging high-capacity cathode materials. However, much current research focuses on the battery materials level, and there have been very few accounts of cell design principles. Here we discuss crucial conditions needed to achieve a specific energy higher than 350 Wh kg−1, up to 500 Wh kg−1, for rechargeable Li metal batteries using high-nickel-content lithium nickel manganese cobalt oxides as cathode materials. We also provide an analysis of key factors such as cathode loading, electrolyte amount and Li foil thickness that impact the cell-level cycle life. Furthermore, we identify several important strategies to reduce electrolyte-Li reaction, protect Li surfaces and stabilize anode architectures for long-cycling high-specific-energy cells.
Lithium (Li)-ion batteries have had a profound impact on modern society1. Over the past 25 years, the specific energy of Li-ion batteries has steadily increased while their cost has dramatically decreased. However, the electric vehicle energy-storage market demands an even higher specific energy, to more than 500 Wh kg−1 at the cell level, and a lower cost, below US$100 (kWh)−1 at the pack level2. At present, many approaches are being pursued in developing next-generation high-energy batteries, such as Li-oxygen and Li-sulfur batteries2,3,4. All solid-state batteries also have received widespread attention due to their inherent high safety characteristics5. Among these approaches, Li metal is considered a key component to achieving a higher specific energy than that of today’s Li-ion technology. Most studies so far have focused on characterization and mitigation of dendrite formation on the material level6,7,8. Although there have been some studies on new cell architectures with improved performances9 and on properties of pouch cells10, there have been very few reports on how the many new materials and concepts studied in the literature should be incorporated into a practical high-energy cell with specific energy higher than 300 Wh kg–1 and what is needed to improve the cell-level energy density and cycle life of practical rechargeable Li metal batteries.
Here we provide a cell-level analysis of what we consider to be the crucial conditions for a rechargeable Li metal battery to achieve a specific energy higher than 350 Wh kg−1, up to 500 Wh kg−1, using a high-capacity cathode such as a high-nickel-content lithium nickel manganese cobalt oxide (high-Ni NMC) (Ni ≥ 60%) based on a pouch-cell format. We discuss the cell-level parameters required to increase the cell energy and cycle life. Rather than providing a complete review or summary of the properties of and advances on the Li metal anode, cathode or electrolyte, we highlight the factors that cause the cell to fail in short cycle life and limit the efficient utilization of the full capacity of the active materials, and identify key strategies to extend the cycle life of the full cells.
Material requirements for 500 Wh kg–1 practical cells
To develop the most feasible approach to build the highest-specific-energy cells, we consider the best cathode and anode candidates that are available in large quantities for cell fabrication at an industrial scale. Among the known anode materials, Li metal has an ultrahigh theoretical specific capacity (3,860 mAh g−1) and an extremely low standard electrochemical redox potential (−3.040 V versus the standard hydrogen electrode), and therefore is considered one of the most important anode materials for future energy storage systems11. Among the reported lithium intercalation cathode materials1, we select high-Ni NMC (LiNixM1−xO2, M = Mn, Co and x ≥ 0.6) based on the consideration of its capacity (greater than 200 mAh g–1), operating voltage (ca. 3.8 V) and commercial availability. Other emerging cathode materials can be explored in the future for even higher specific capacities, including sulfur or oxygen3,4, metal fluoride materials12, lithium-rich manganese-rich layered oxide materials13 and anion redox cathode materials14.
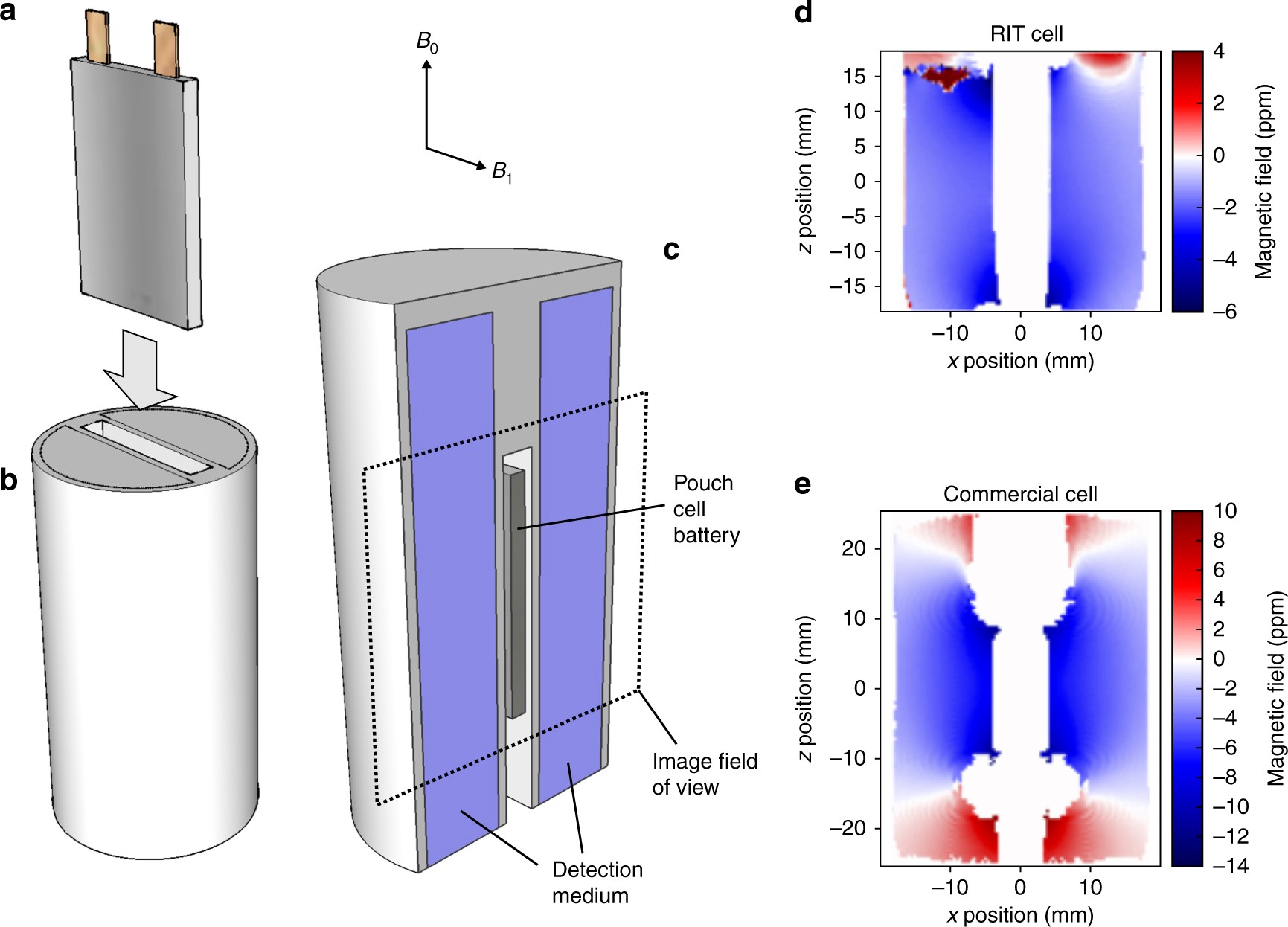
A commercially viable cell needs to meet many requirements, including high specific energy, long cycle life, good mechanical and chemical stability over a wide range of temperature, safe operation, and so on. As a starting point, this Perspective analyses the crucial conditions of the cell parameters required for a high-energy cell using a Li||high-Ni NMC (>1 Ah) pouch-cell format containing multiple layers of current collectors, anodes, separators and cathodes. The pouch cell is chosen because it is one of the most common cell formats in commercial production with high manufacturing flexibility.
To integrate the best material properties with the optimal cell design parameters to achieve a specific energy up to 500 Wh kg–1, we plotted the cell-level specific energy under different scenarios to illustrate the feasible pathways to different targets (Fig. 1). The cell-level specific energy is calculated by multiplying the total cathode capacity and the cell voltage (taken as the mid-point voltage in discharge), and then dividing by the total mass of the pouch cell. For the baseline cell, parameters similar to a typical Li-ion battery are used. These baseline parameters, including the cathode thickness, porosity, amount of electrolytes and so on, represent what can be reasonably implemented in the pouch-cell fabrication based on knowledge from Li-ion cells and thus serve as a starting point for our analysis. The challenge is to optimize these parameters to reach the desired specific energy while still achieving good cycle life in practical Li metal cells. Figure 1 shows the upper limit of the specific energy when those key parameters are pushed to the limit.
You can see full article on:
https://www.nature.com/articles/s41560-019-0338-x